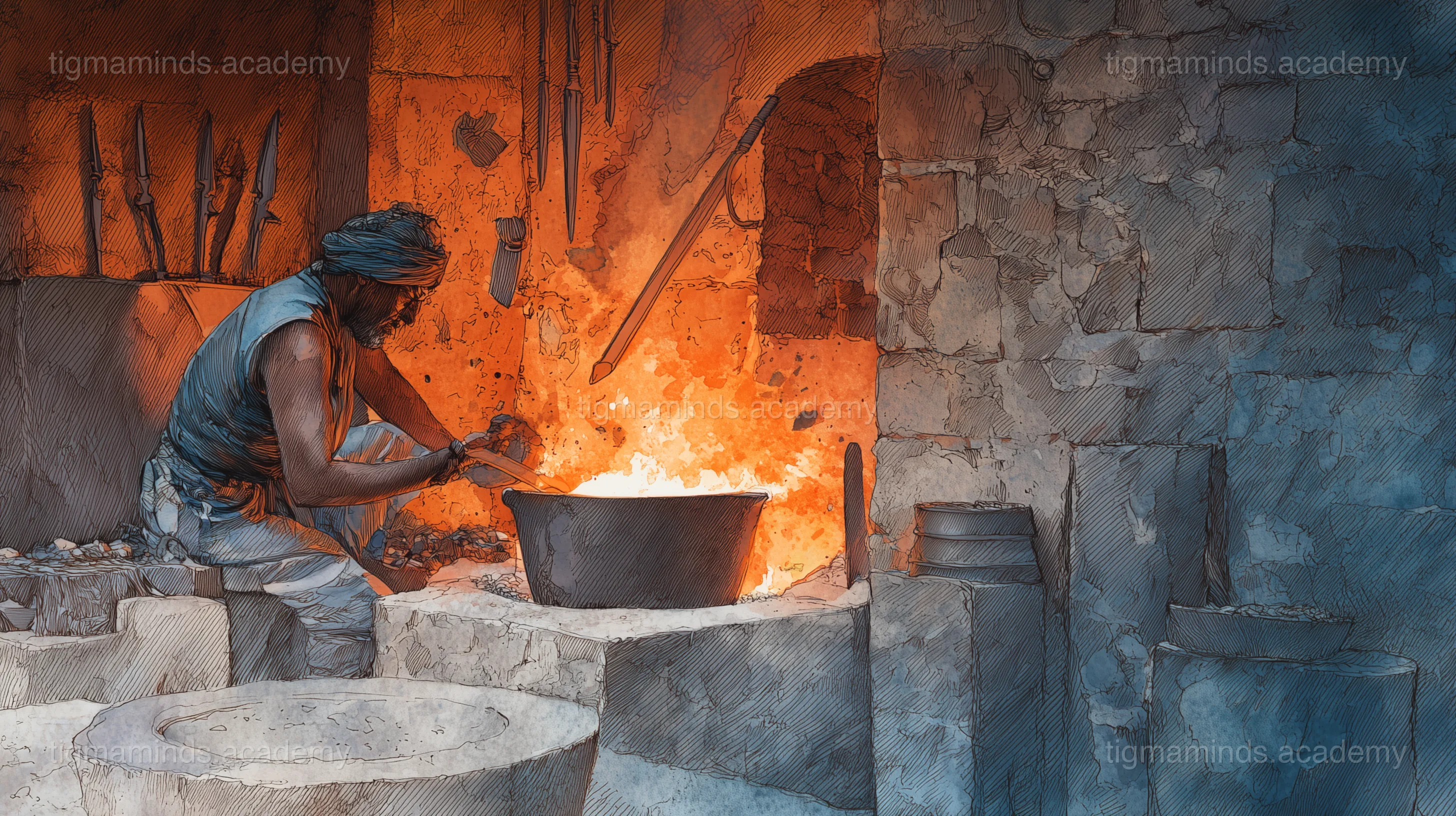
Indian Wootz Steel
The legendary Damascus steel — carbon nanostructures discovered 2,000 years before nanotechnology.
The Story
The Sword That Cut Silk
In the year 1192, during the Third Crusade, a legend circulated among the Crusader armies about the swords carried by Saladin's warriors. These blades, the stories said, could cut a silk scarf dropped onto the edge — the weight of the falling fabric alone was enough to part it. They could cleave through European swords that were twice as thick. And their surfaces showed a distinctive pattern of light and dark bands, like flowing water — the mark of Damascus steel.
The Crusaders were terrified of these blades. They tried to buy them, steal them, and capture the smiths who made them. They failed on all counts. Because the secret of Damascus steel was not in Damascus. It was in India.
Crucible Steel
The raw material for Damascus blades was wootz — a type of steel produced in crucibles in southern India, particularly in the regions now known as Tamil Nadu, Karnataka, and Telangana. The process had been developed as early as 300 BCE, making it one of the oldest steelmaking techniques in the world.
The method was deceptively simple. Iron ore was mixed with charcoal and sealed in a small clay crucible — a pot about the size of a football. The crucible was placed in a charcoal-fired furnace and heated to approximately 1,200°C — hot enough to melt the iron and dissolve the carbon from the charcoal into the molten metal.
Ordinary iron contains very little carbon (less than 0.1%). Steel is iron with 0.5-2% carbon. The carbon atoms fit into the gaps between iron atoms in the crystal lattice, making the metal harder and stronger. But controlling the carbon content — getting exactly the right amount — requires precise control of temperature, time, and atmosphere.
The Indian crucible method achieved this control beautifully. The sealed crucible prevented oxygen from reaching the molten metal (which would burn off the carbon). The small volume of the crucible ensured even heating. And the specific ratio of iron to charcoal, refined over centuries of practice, produced steel with a carbon content of approximately 1.5% — the ideal range for a blade that is both hard (it holds an edge) and tough (it doesn't shatter on impact).
The Pattern
The distinctive wavy pattern of Damascus steel — called the damask pattern — is formed during the slow cooling of high-carbon crucible steel. As the metal cools, the carbon atoms segregate into bands of cementite (iron carbide, Fe₃C) — a very hard, brittle compound — embedded in a matrix of softer, more ductile iron.
The cementite bands are microscopically thin — just a few micrometres across. But there are millions of them, layered through the blade. When the smith forges the ingot into a blade (heating and hammering repeatedly), these bands deform and flow, creating the visible swirling pattern on the surface. Etching the finished blade with weak acid reveals the pattern, because the cementite bands resist the acid differently than the surrounding iron.
In 2006, researchers at the Technical University of Dresden examined samples of genuine Damascus steel using electron microscopy and discovered something extraordinary: the cementite bands contained carbon nanotubes — cylindrical structures of carbon atoms just a few nanometres in diameter.
Carbon nanotubes are one of the strongest materials known to science. They have a tensile strength 100 times greater than steel at one-sixth the weight. They are a product of 21st-century nanotechnology — or so we thought. The Indian steelmakers were producing them, unknowingly, 2,000 years before the word "nanotechnology" was coined.
The nanotubes formed because of trace impurities in the Indian iron ore — small amounts of vanadium, molybdenum, and tungsten — that catalysed the formation of carbon nanostructures during the slow cooling process. The Indian smiths didn't know about nanotubes. But they knew which ores produced the best steel, and they passed that knowledge down through generations.
The Lost Recipe
By the mid-18th century, the production of true wootz steel had ceased. The specific ores that contained the right trace impurities were exhausted. The family workshops that had guarded the technique for centuries were disrupted by colonial rule. The knowledge was lost — not suddenly, but gradually, as the chain of master-to-apprentice transmission was broken.
Despite two centuries of effort by metallurgists — including Michael Faraday, who published a study on Indian steel in 1819 — no one has been able to fully reproduce wootz steel with all its properties. Modern metallurgy can create steel with the right carbon content and even produce similar surface patterns. But the exact combination of ore chemistry, crucible design, firing temperature, cooling rate, and forging technique that produced the original blades — with their carbon nanotubes and their silk-cutting edges — remains elusive.
What It Teaches
Wootz steel is a humbling lesson for modern science. We assume that advanced materials are products of advanced understanding — that you need electron microscopes and particle accelerators to make nanostructured materials. But Indian steelmakers produced carbon nanotubes in clay pots over charcoal fires, guided by nothing but empirical knowledge — the accumulated observations of hundreds of generations of smiths who noticed which ores, which temperatures, and which cooling rates produced the best blades.
Science is not only what we understand theoretically. It is also what we discover through practice — through doing, observing, refining, and passing on. The Indian smiths were scientists. They just didn't call themselves that.
The end.
Before you start
Try It Yourself
Choose your level. Everyone starts with the story — the code gets deeper as you go.
Story Progress
0%Ready to Start Coding?
Here is a taste of what Level 1 looks like for this lesson:
import numpy as np
import matplotlib.pyplot as plt
# Your first data analysis with Python
data = [45, 52, 38, 67, 41, 55, 48] # measurements
mean = np.mean(data)
plt.bar(range(len(data)), data)
plt.axhline(mean, color='red', linestyle='--', label=f'Mean: {mean:.1f}')
plt.xlabel("Sample")
plt.ylabel("Value")
plt.title("Metallurgy & Materials Science — Sample Data")
plt.legend()
plt.show()This is just the first of 6 coding exercises in Level 1. By Level 4, you will build: Build a Steel Properties Calculator.
Free
Level 0: Listener
Stories, science concepts, diagrams, quizzes. No coding.
You are here
Level 0 is always free. Coding levels (1-4) are part of our 12-Month Curriculum.